
This New Oral Vaccine Could Slash E.coli Diarrhea Cases by Over 80%—Are You at Risk?
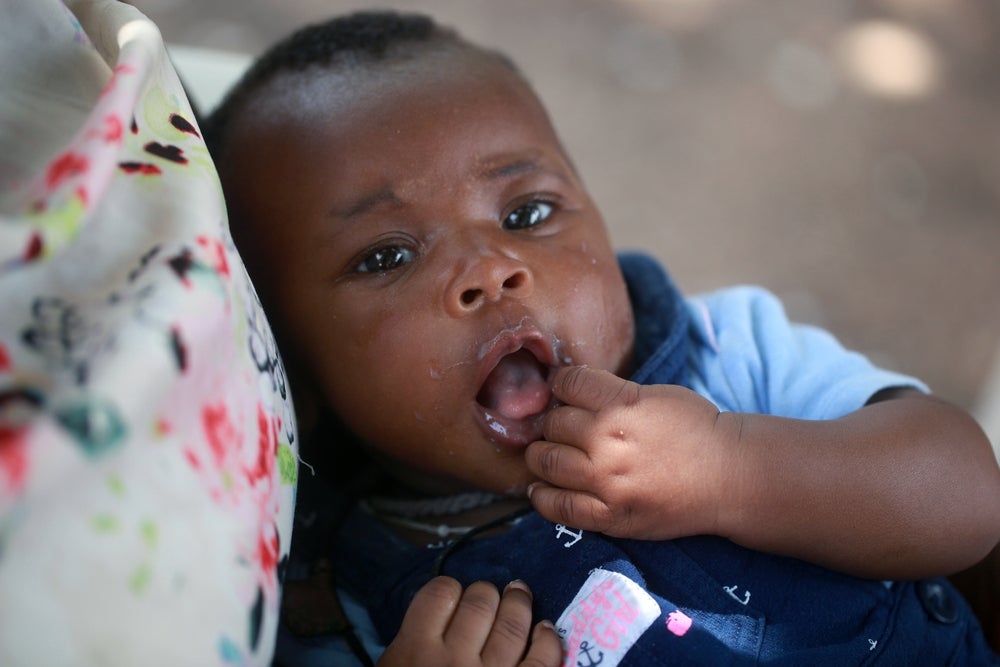
A novel oral vaccine, ETVAX, has shown promising results in reducing cases of moderate-to-severe diarrhoea caused by enterotoxigenic E. coli (ETEC) in children, according to a Phase IIb study. Conducted in Gambia and funded by the European & Developing Countries Clinical Trials Partnership (EDCTP2), the trial demonstrated an efficacy rate of up to 80.6% in a subset of participants.
The study enrolled 4,936 children aged six to 18 months, who were randomly assigned to receive three doses of either ETVAX or a placebo on days 1, 15, and 90 of the study. The primary endpoint was to assess the vaccine's efficacy against ETEC-positive diarrhoea in children not co-infected with other pathogens such as rotavirus, norovirus, Shigella, or Cryptosporidium.
While ETVAX did not meet its primary endpoint—showing an efficacy rate of only 26.6% (p=0.43)—it narrowly missed achieving statistical significance when evaluating its impact against ETEC without regard to co-infections, recording an efficacy of 48.2% (p=0.053). However, on secondary and exploratory endpoints, the vaccine exhibited substantial promise, particularly in children who were not infected with specific intestinal parasites.
This study emphasized the importance of early administration of ETVAX, revealing that 67.8% of children who began their vaccination course before the age of nine months were protected against ETEC-positive diarrhoea. Additionally, the vaccine's safety profile was comparable to that of the placebo, with serious adverse events (SAEs) occurring in 1% (24) of the ETVAX group and 1.3% (32) of the placebo group, with none related to the vaccine itself.
The Path Forward for ETVAX
According to Thomas Wierzba, the corresponding author of the study published in The Lancet, this represents the first instance of an oral ETEC vaccine showing potential to provide "real protection in a high-burden, low-income setting." Following the promising results from the Phase IIb study, the European Medicines Agency (EMA) has approved the advancement of ETVAX to a pivotal Phase III trial. This upcoming trial will assess the vaccine's consistency across different populations.
If successful, ETVAX could become the first vaccine to protect against ETEC-induced diarrhoea, a condition that currently accounts for an estimated 42,000 deaths annually. The vaccine also demonstrated a 21% reduction in the overall prevalence of moderate-to-severe diarrhoea of all causes at two years, positioning it as a critical addition to diarrhoea treatment strategies, especially in low-income regions.
Should ETVAX gain regulatory approval, it would join the ranks of existing vaccines like MSD’s RotaTeq and GSK’s Rotarix, which are already in use to protect against common rotaviral infections causing diarrhoea. UNICEF plays a significant role in providing these vaccines to children in low-to-middle income countries, enhancing public health outcomes.
In a related development, Moderna is also investigating a potential mRNA-based vaccine for norovirus, termed mRNA-1403, which is currently in a Phase III trial that began dosing in 2024.
The advancements made with ETVAX signal a hopeful future in addressing diarrhoea-related illnesses in vulnerable populations, potentially transforming health outcomes for children globally.



You might also like: